Welcome to the first entry in our Electronics Basics series! To understand how your phone, computer, or even a simple flashlight works, you must first get to know the “star” of the show: the electron. Without the movement of these tiny particles, electronic technology as we know it simply would not exist.
1. Atomic Structure: The Electron’s “Home”
All matter in the universe is made of atoms. To understand electrons, we often use the analogy of a mini solar system, though modern science gives us a more detailed picture.
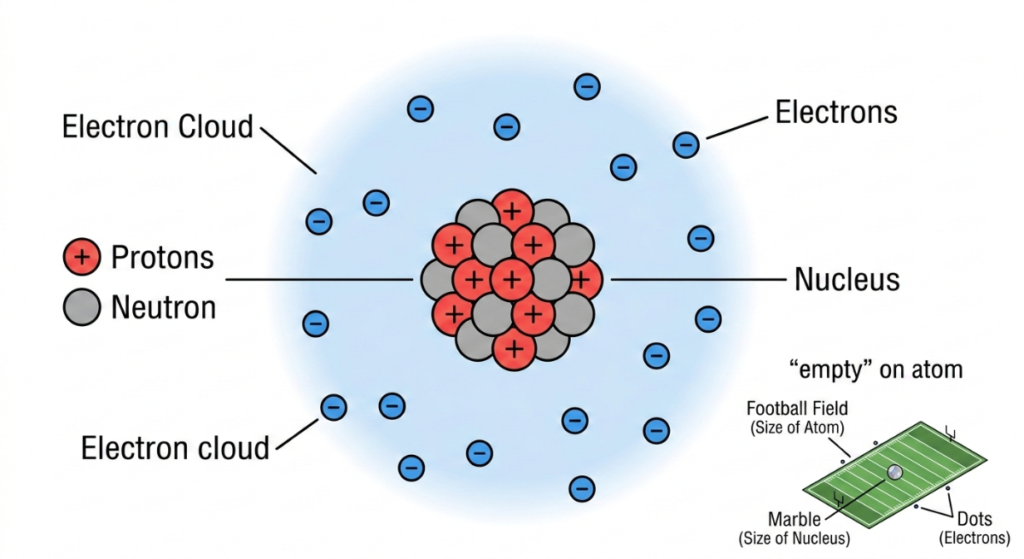
- The Nucleus: At the center of the atom is a dense core called the nucleus. It contains protons, which have a positive (+) electrical charge, and neutrons, which have no charge. The number of protons determines what element the atom is (for example, a copper atom always has 29 protons).
- The Electron Cloud: Electrons are tiny, lightweight particles that carry a negative (-) electrical charge. While we often visualize them orbiting the nucleus like planets, they actually move at incredible speeds within an area called the electron cloud.
- The Power of Empty Space: Interestingly, an atom is mostly empty space. If the nucleus were the size of a marble in the center of a football field, the electrons would be like tiny gnats buzzing around the very edges of the stadium.
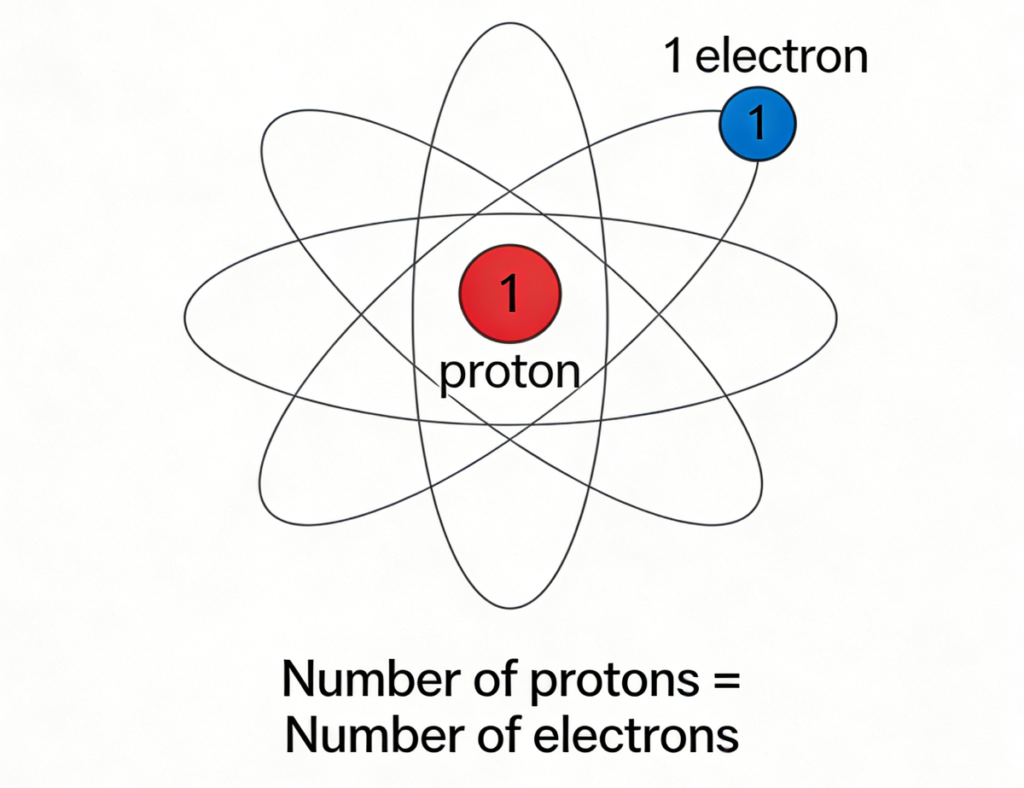
In a neutral atom, the number of protons equals the number of electrons, meaning the positive and negative charges are perfectly balanced.
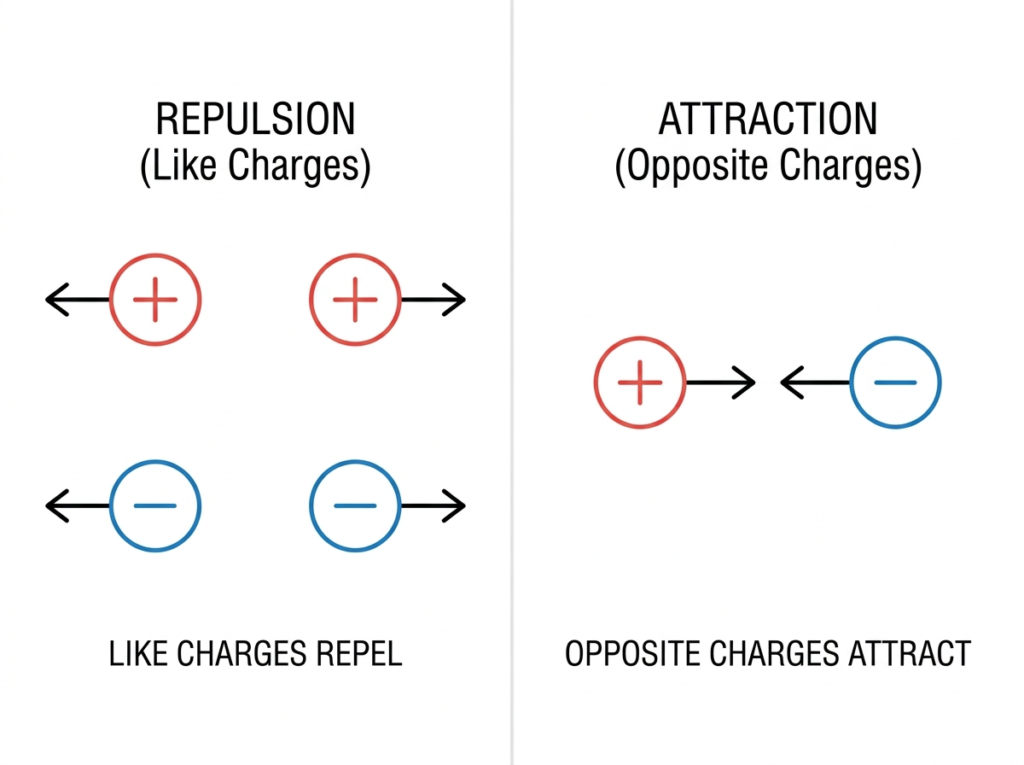
2. Electrical Charge: The Motivation for Movement
Charge is a fundamental property of matter. The most important rule to remember in electronics is: Like charges repel each other, and opposite charges attract. This attraction between the positive nucleus and the negative electrons is what usually keeps an atom together.
To measure electrical charge, we use a unit called the Coulomb (C). Because electrons are so small, it takes a staggering amount of them to equal one Coulomb—approximately 6.24×1018 electrons (that is 6.24 billion billion!).
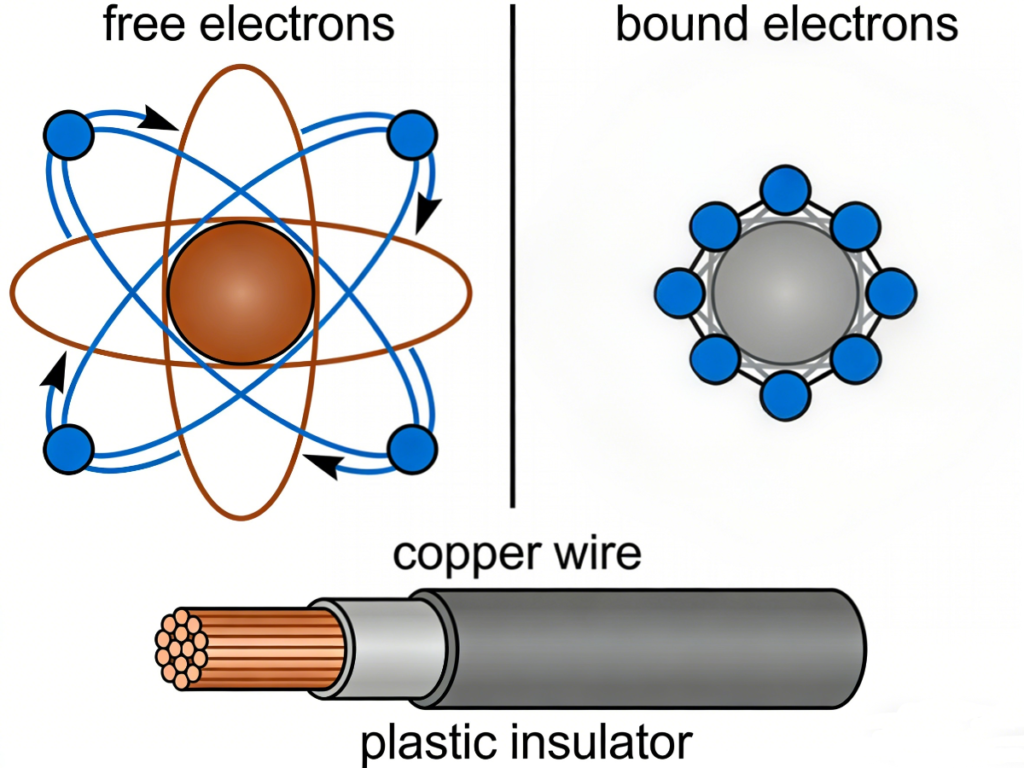
3. Conductors, Insulators, and “Free Electrons”
Not all materials treat their electrons the same way, which is why some things conduct electricity while others don’t:
- Conductors: In metals like copper, silver, and aluminum, the atoms hold their outermost electrons very loosely. These are called free electrons because they can easily break away and move from one atom to another.
- Pro Tip: Silver is actually the best conductor (with a relative conductivity of 1.064), but copper (1.000) is the industry standard because it is much more affordable while still being an excellent conductor.
- Insulators: Materials like rubber, plastic, and glass hold their electrons very tightly. Because there are almost no free electrons to move around, electricity cannot flow through them. This is why we wrap copper wires in plastic—to keep the electrons flowing inside the wire and not into you!
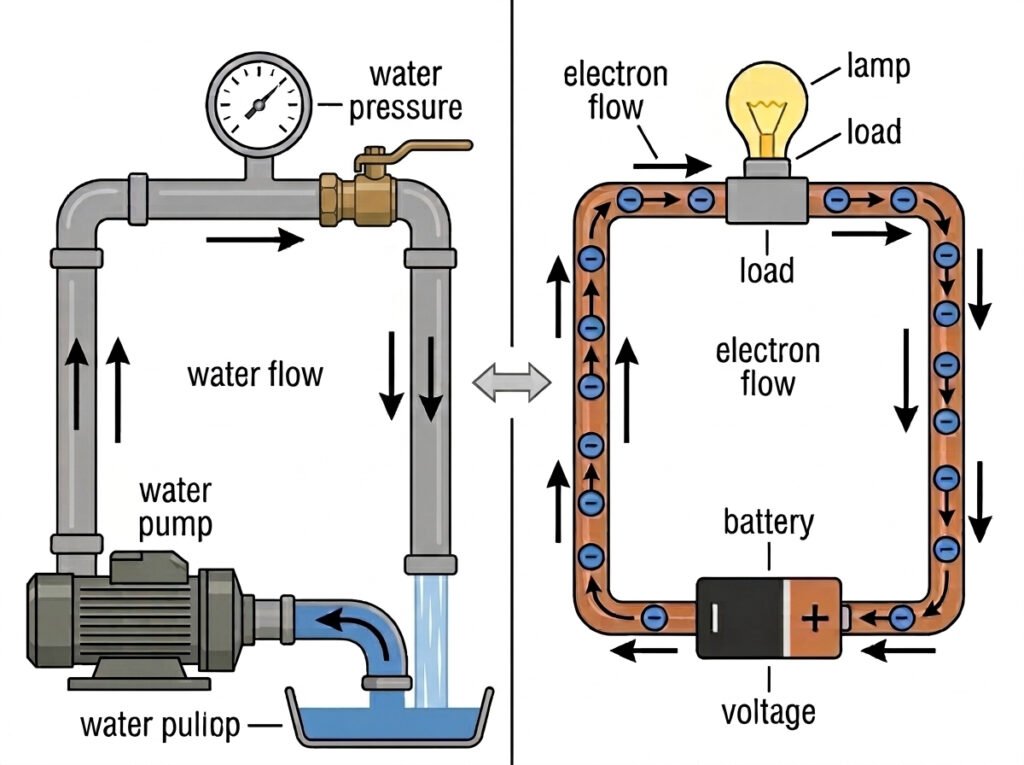
4. How Electron Flow Becomes Current
How do we get these free electrons to move in a useful way? We need an external force, such as a battery. When a battery is connected to a circuit, it creates an electric field through the wire. This field exerts a push on the free electrons. You can visualize this using the
Water Pipe Analogy:
- Current is like the flow of water through the pipe. In a wire, current is the directed flow of billions of electrons moving in the same direction.
- Voltage is like water pressure. It is the “push” provided by the electric field that forces the electrons to move.
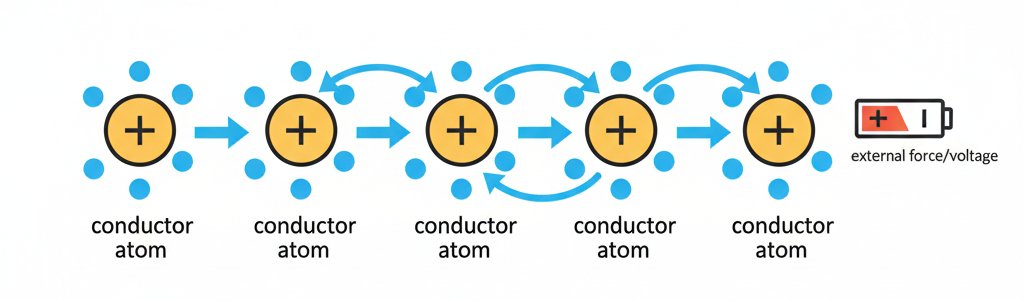
As one electron is pushed out of its “home” atom, it moves to a neighbor, which in turn knocks another electron loose. This chain reaction, happening at near the speed of light, is what we call electric current.
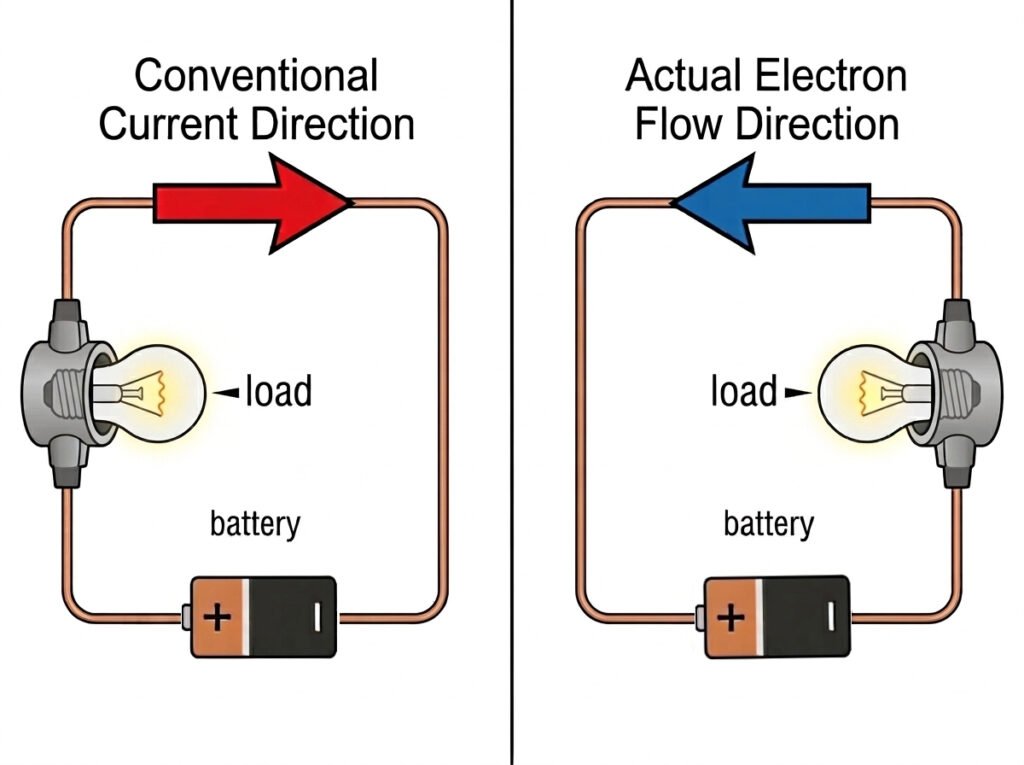
5. A Historical Quirk: Current Direction
There is one confusing “historical quirk” you will encounter in electronics. In the 18th century, Benjamin Franklinassumed that electricity flowed from positive (+) to negative (-). We call this conventional current, and it is still used in almost all circuit diagrams today.
However, we now know that electrons actually flow from negative to positive. Don’t let this worry you—just remember that the electrons are the physical particles moving, even if we draw the arrows pointing the other way!
6. Summary:
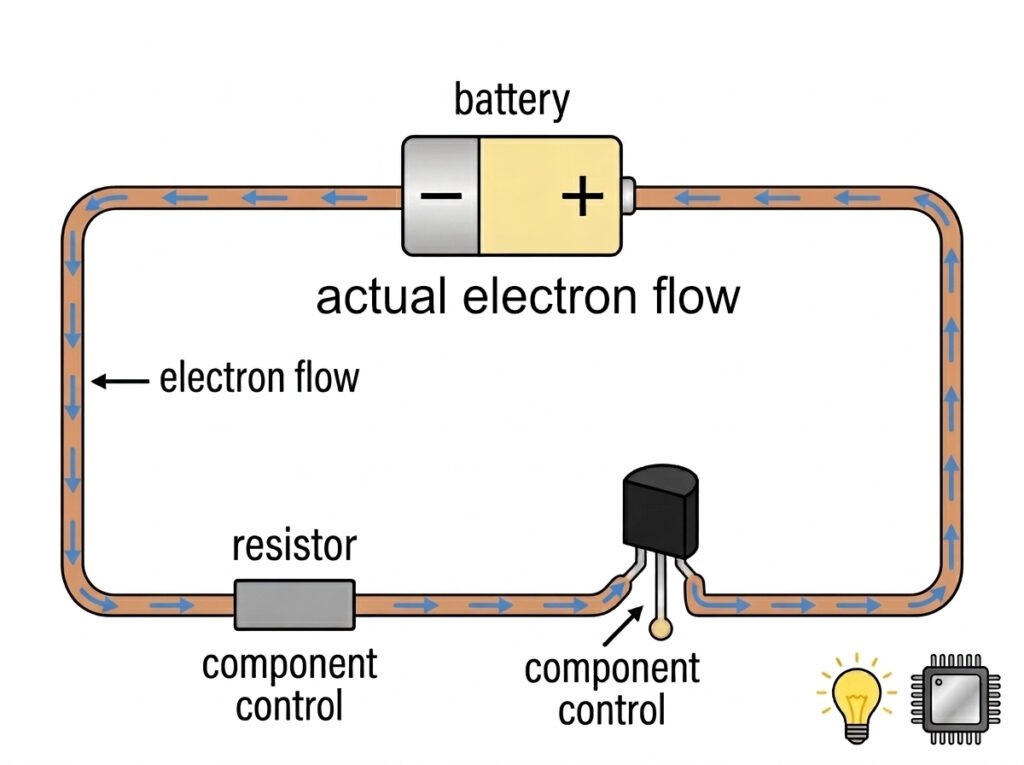
In short, electronics is the science of controlling the flow of electrons. By using components like resistors and transistors, we can guide, slow down, or stop these tiny negatively charged particles to make them do amazing things—from lighting up a bulb to processing data in a microchip.
Next Up in Electronics Basics: Now that we know what is moving, our next article will explore electric charges, static charge, and currency!