Welcome back to our Electronics Basics series! In the last lesson, we got to know electrons—the tiny, negatively charged particles that make all electronic devices work. Today, based on that foundation, we’ll explore three core concepts: electric charge, static charge, and current. By the end of this article, you’ll understand the key concepts of these terms, knowing how they behave in everyday electronics.
1. Electric Charge: The “Push” Behind Electricity
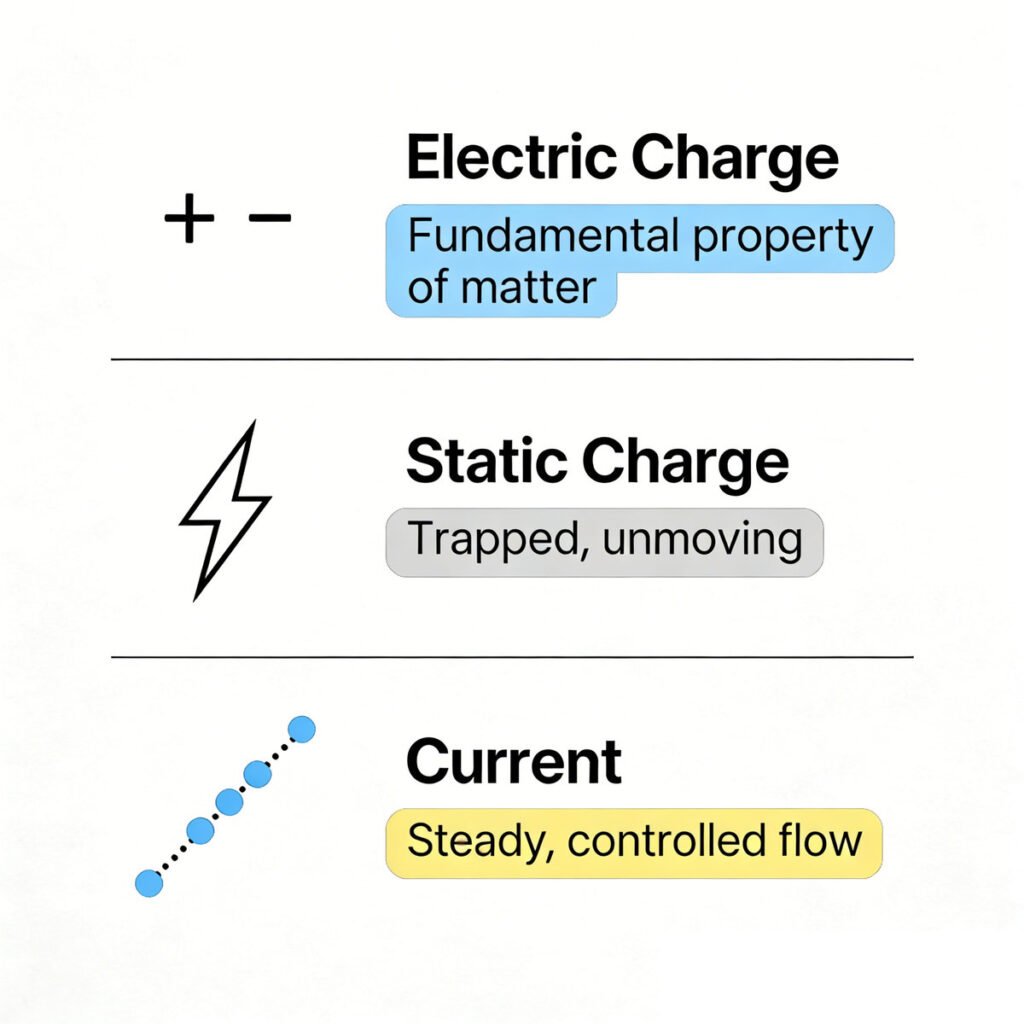
We briefly mentioned electric charge in the last lesson, but let’s break it down in simple, easy-to-understand terms: electric charge is a fundamental property of matter, just like mass (how heavy something is) or volume (how much space something takes up). It is the primary reason electrons and protons interact with each other.
There are only two types of electric charge in the entire universe, and they follow one unbreakable rule:
- Positive charge (+): This type of charge is found in protons, which are the tiny particles located in the nucleus (the center) of every atom. Protons never move from one atom to another—they stay fixed in the nucleus, so the positive charge of an atom is always constant (unless the atom is split, which is not something that happens in everyday electronics).
- Negative charge (-): This type of charge is found in electrons, the tiny particles that orbit the nucleus of an atom (like planets orbiting the sun). Unlike protons, electrons are loosely held by nucleus (especially in conductors like copper), so they can move from one atom to another—and this movement is what creates electricity.
- Like charges repel each other (positive ↔ positive, negative ↔ negative): Just like two north poles of a magnet push each other away, two particles with the same charge will repel (push away) each other. For example, two electrons (both negative) will push each other apart, and two protons (both positive) will do the same.
- Opposite charges attract each other (positive >< negative): This is the “pull” force—just like a north and south pole of a magnet pull together, a positive proton and a negative electron will attract each other. This attraction is what keeps electrons orbiting the nucleus of an atom, and it’s also what makes electrons move from one atom to another.
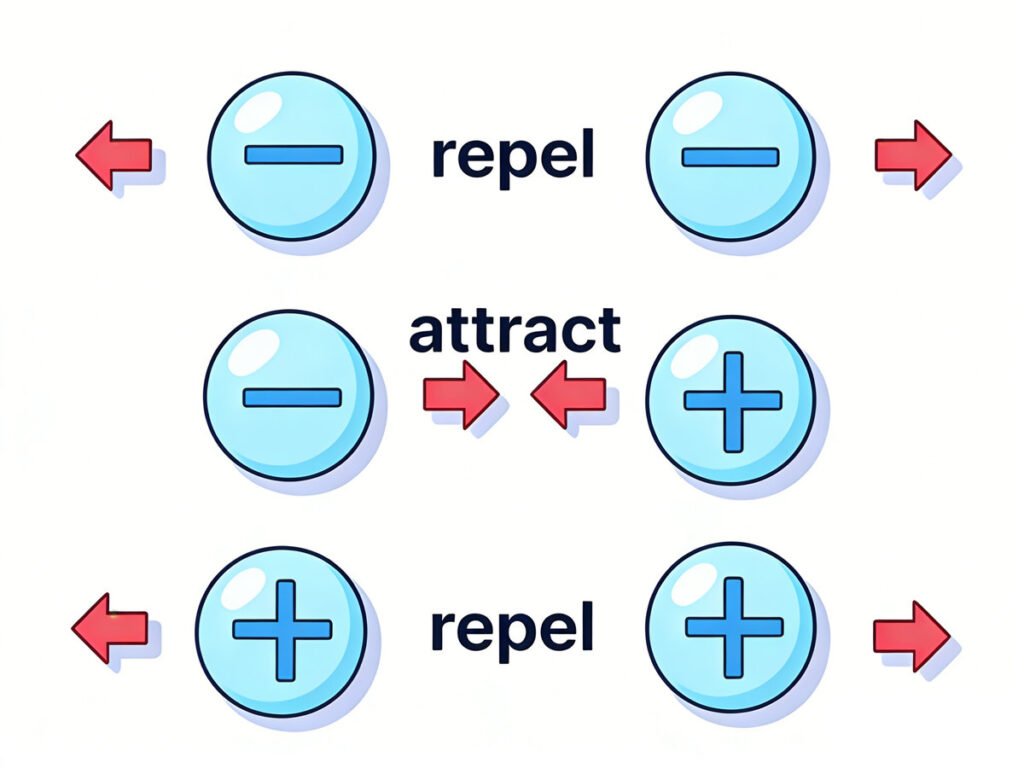
Think of it like magnets: two north poles push each other away, while a north and south pole pull together. Electric charge works exactly the same way—this attraction and repulsion between charged particles is what makes electrons move, and moving electrons are the basis of all electricity we use every day.
In a neutral atom (an atom with no overall charge), the number of positive protons in the nucleus equals the number of negative electrons orbiting the nucleus. This means the positive and negative charges cancel each other out—so the atom doesn’t have a positive or negative “charge” overall. But when atoms gain or lose electrons, they become the “charged particles” called ions—and that’s where static charge comes in.
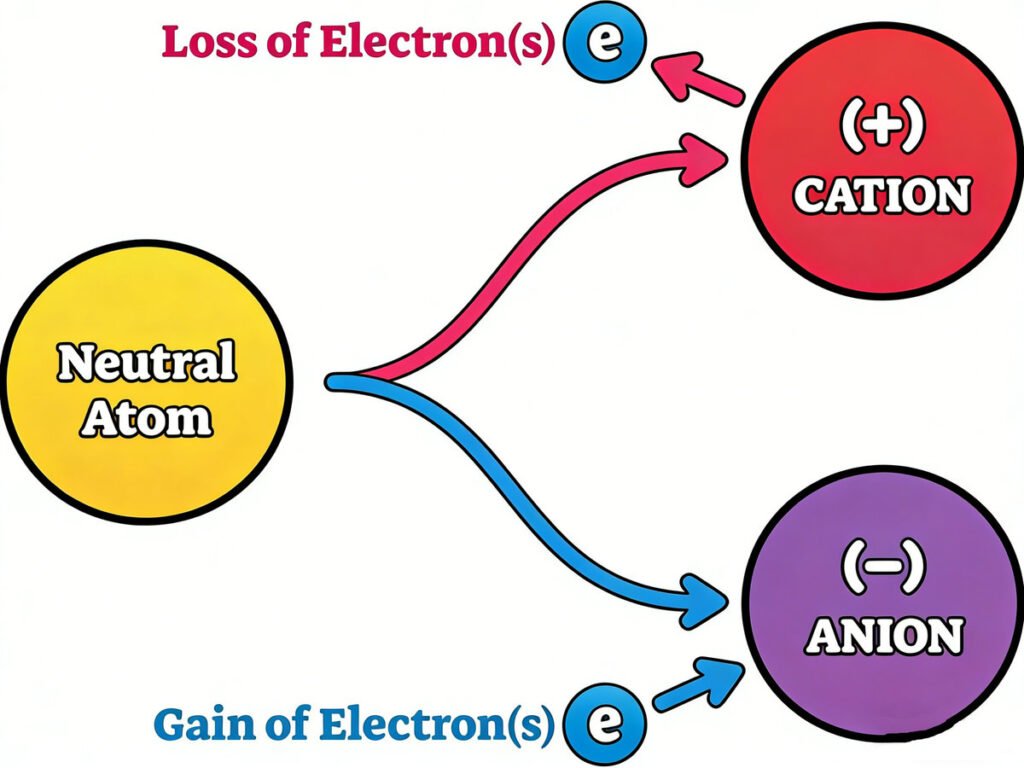
2. Static Charge: Trapped, Unmoving Charge
You’ve probably experienced static charge before, even if you didn’t know what it was: when you rub a balloon on your hair and it sticks to a wall, or when you walk across a carpet and then touch a doorknob, only to feel a tiny, harmless spark. That’s the “static charge” in action—and it’s easier to understand than you might think!

Static charge (or static electricity) is simply charge that’s trapped and not moving. It forms when electrons are transferred from one object to another—usually through friction (like rubbing two objects together) or direct contact. Unlike the electricity that powers your phone (which is moving charge), static charge is “stuck” on an object, waiting to be discharged (released) to something else.
The key thing to remember about static charge is that it’s stationary (not moving). The electrons don’t flow in a steady stream like they do in a wire—they just sit on the object, waiting to be discharged (like the spark). This is the big difference between static charge and current, which is moving charge (we’ll get to current next).
Fun fact: Static charge is why lightning happens! Clouds rub against each other (or against the ground) as they move through the sky, which transfers electrons and builds up huge amounts of static charge. When the charge gets too big to stay trapped, it discharges as a lightning bolt—nature’s giant spark! Lightning is just static charge on a massive scale.

3. Current: Moving Charge (The “Flow” of Electricity)
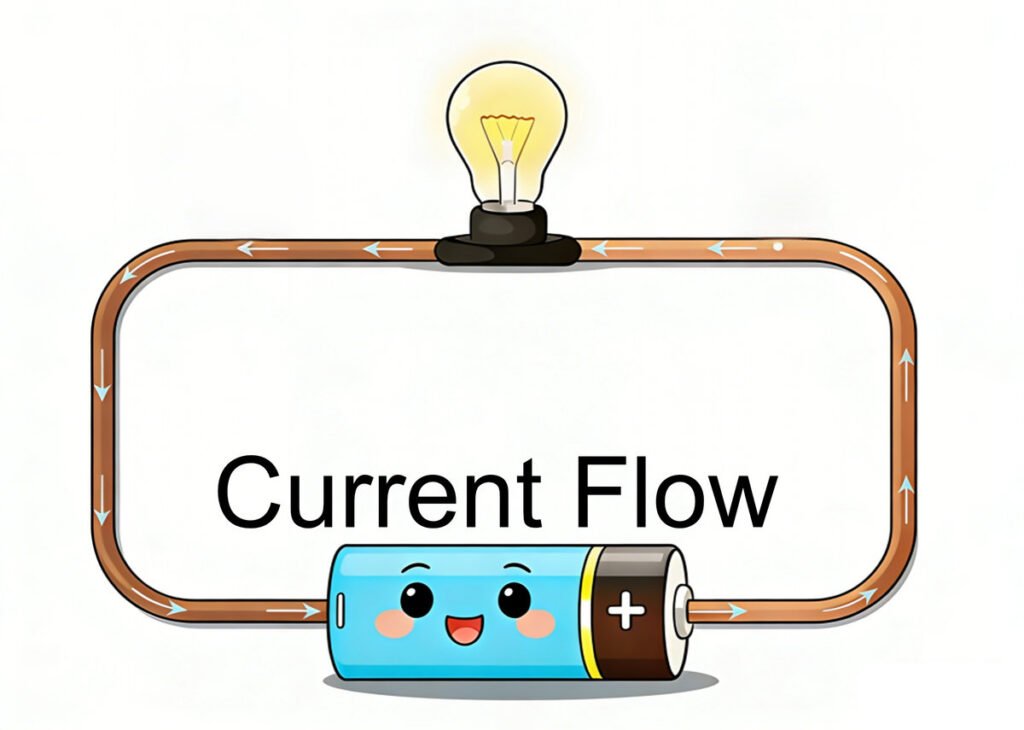
The static charge is trapped, unmoving charge, but current is the charge in motion—specifically, it’s the steady, controlled flow of electrons through a material (like a copper wire). This is the electricity that powers your phone, laptop, flashlight, radio, and every other electronic device you use. Without current, none of these devices would work.
Remember the free electrons we talked about in Lesson 01? In conductors (materials like copper, aluminum, or silver), these electrons are loosely held by atoms—they’re not stuck orbiting one nucleus. Instead, they can move freely from one atom to another within the material. When we apply an external “push” (like a battery), these free electrons start moving in the same direction—and that steady movement is what we call current.
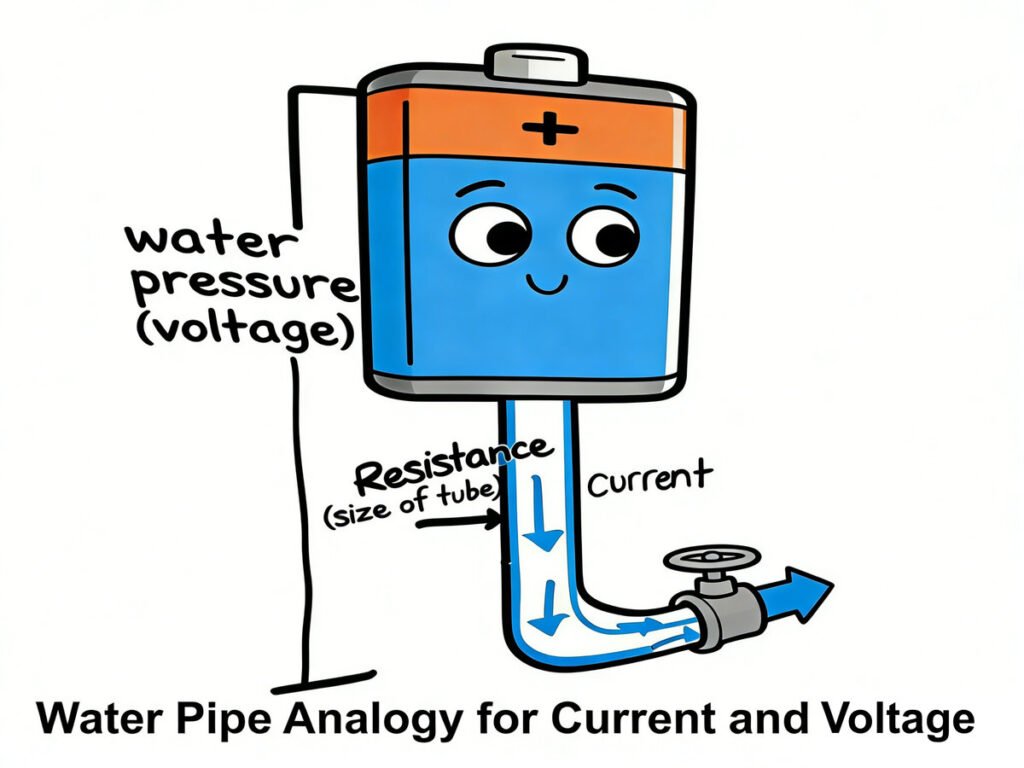
Let’s go back to the Water Pipe Analogy (from Lesson 01) to make current even easier to understand:
- Current = Water flow: Just like water flows through a pipe, electrons flow through a wire. The more electrons that flow through the wire per second, the higher the current. For example, a small flashlight has a low current (not many electrons flowing), while a hair dryer has a high current (lots of electrons flowing).
- Voltage = Water pressure: Voltage is the “push” that makes the electrons (or water) move. A battery provides this push—think of it like a water pump that pushes water through a pipe. The higher the voltage (like a bigger pump), the more “push” there is, and the faster the electrons flow (higher current).
Unlike static charge (which is stationary, unsteady, and trapped), current is a controlled, steady flow of electrons. For current to exist, we need two things: a conductor (a material that lets electrons flow freely, like copper wire) and a voltage source (like a battery or wall outlet) to push the electrons. If you have a wire (conductor) but no battery (voltage source), no electrons will flow—there’s no “push” to make them move. If you have a battery but no wire, the electrons can’t flow—there’s no path for them to take.
A quick note on the unit for current: We measure current in Amperes (A), which is often shortened to “amps” for short. One ampere is the flow of one Coulomb of charge per second. Remember from Lesson 01 that 1 Coulomb = 6.24×10¹⁸ electrons—that’s 6.24 followed by 18 zeros! To put that in perspective, 1 amp is enough to power a small light bulb, while most household outlets provide 10-20 amps (enough to power larger devices like TVs or microwaves).
4. Key Differences: Charge, Static Charge, & Current
It’s easy to mix these three concepts up, especially since they’re all related to electricity. Let’s summarize their differences in simple terms, with extra explanations to make sure you don’t confuse them:
| Concept | What It Is (Simplified) | Key Trait (Easy to Remember) | Everyday Example |
| Electric Charge | A fundamental property of matter—like mass or volume—that makes particles attract or repel each other. It comes in two types: positive (+) and negative (-). | Exists in all atoms; it’s the “source” of electricity (without charge, there’s no electricity). | The charge in a proton (positive) or electron (negative). |
| Static Charge | Charge that’s trapped on an object and not moving. It forms when electrons are transferred (via friction or contact) and get stuck. | Unsteady; it discharges as a spark (no continuous flow of electrons). | Paper scraps sticking to a plastic rod, or a shock from a doorknob. |
| Current | Steady, controlled flow of electrons through a conductor (like a wire), powered by a voltage source (like a battery). | Continuous and controlled; it powers electronic devices. | The electricity that flows through a phone charger to power your phone. |
5. Summary: Charge → Static Charge→ Current
To wrap it up in simple terms, let’s connect all three concepts so you can see how they work together: Electric charge is the basic property that makes electrons and protons interact (attract or repel). Static charge is when that charge gets trapped on an object and doesn’t move (like the plastic rod and paper scraps example). Current is when that charge (specifically, electrons) flows steadily through a conductor—powered by voltage—and this flow is what powers all your electronic devices.
In the next lesson, we’ll dive deeper into voltage, current, and resistance—using the water pipe analogy to make these concepts even easier to understand (no complex formulas, promise!). We’ll also talk about how these three work together in a simple circuit, so you can start connecting the dots between the basics and real-world electronics.

